
Chlorine vs. Chloramine: What's the Difference?
28th Feb 2026
Key Takeaways
- Chlorine is highly effective but dissipates quickly and creates more harmful disinfection byproducts (DBPs).
- Chloramine is a combination of chlorine and ammonia; it is much harder to remove because it does not "boil off" or evaporate like chlorine.
- Standard carbon filters (like most pitchers) are often ineffective against chloramine due to insufficient contact time.
- Catalytic carbon is the scientific gold standard for neutralizing the ammonia bond in chloramines.
- According to the CDC, both are safe at regulated levels, but chloramine is more "persistent" in the distribution system.
If you’ve noticed that your tap water still has a distinct "swimming pool" smell or a metallic chemical taste (even after running it through a standard pitcher or refrigerator filter), you aren’t imagining things. You are likely dealing with chloramine, the stubborn, "sticky" cousin of chlorine that modern municipalities are increasingly using to treat our water supply.
While basic filters are designed to handle simple chlorine, they often fail miserably against the complex chloramine bond. To fix your water, you first have to understand the chemistry of what’s actually in it.
What is Chlorine? The Traditional Standard
For over a century, secondary water disinfection has relied almost exclusively on free chlorine. It is a powerful oxidizer that effectively kills bacteria, viruses, and other pathogens on contact.
However, chlorine has two major flaws. First, it is unstable; it gasses off quickly, meaning water at the end of a long pipeline might not have enough disinfectant left to keep it safe. Second, when chlorine reacts with organic matter in the water, it creates Disinfection Byproducts (DBPs) like trihalomethanes, which have been linked to long-term health risks.
What is Chloramine? The Persistent Successor
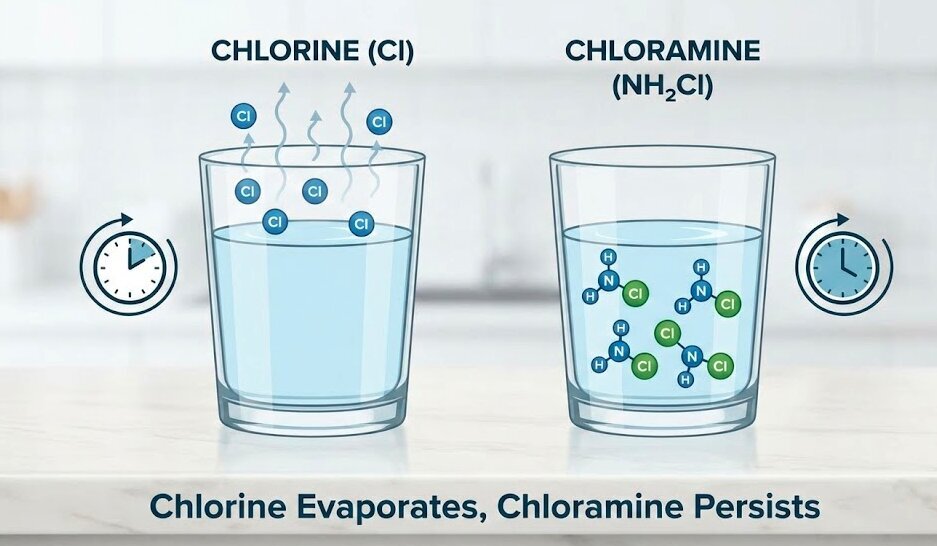
Chloramine is created by adding ammonia in drinking water treatment stages where chlorine is already present. This creates a more stable compound (monochloramine).
Unlike chlorine, chloramine does not dissipate easily. While this is a "pro" for the city (ensuring the water stays disinfected all the way to your tap), it is a "con" for the homeowner. Because it is so stable, you cannot simply leave a jug of water on the counter overnight to let the chemicals evaporate. The ammonia bond holds the chlorine in place, making it a "persistent" chemical.
Why Cities Switched
Many homeowners wonder why their water changed. Most municipalities are switching to chloramine to meet stricter EPA regulations regarding DBPs.
- Longevity: Chloramine stays active in the pipes longer than chlorine.
- Reduced DBPs: Chloramine produces fewer regulated byproducts like THMs.
- Cost-Effectiveness: Because it lasts longer, cities often need to use less of it over long distances.
However, this stability comes with a trade-off. As noted by the EPA, while chloramines are safe for drinking and bathing, they can be significantly more corrosive to certain types of plumbing (like copper or lead) if the water chemistry isn't perfectly balanced.
The Filtration Problem: Why Basic Filters Fail
If you are comparing chlorine vs. chloramine water filters, you’ll find that standard Activated Carbon (AC) is great for chlorine but poor for chloramine.
Removing chloramine requires a chemical reaction to break the chlorine-ammonia bond. This requires significantly more contact time, the amount of time the water spends touching the filter media. A standard "drop-in" carbon filter simply doesn't have the surface area or the specialized chemistry to do the job before the water rushes through to your glass.
A standard carbon filter might remove 95% of chlorine but as little as 20% of chloramine at the same flow rate.

How to Remove Chloramines from Tap Water
To truly "rescue" your water from that chemical taste, you need equipment designed for the specific molecular structure of chloramines.
1. Catalytic Carbon for Chloramine
Unlike standard carbon, catalytic carbon is a high-performance media that has been structurally altered to promote the chemical reaction needed to break the ammonia bond. It is the only reliable way to ensure the "chlor" and the "amine" are separated and neutralized.
2. Increased Contact Time
Because the reaction takes longer, you need a larger tank system. This is why "whole-home" solutions are superior to under-sink versions. Systems like the Manor Duo provide the necessary housing size to ensure the water isn't just passing through, but is actually being treated.
3. Comprehensive Filtration
Chloramine is often found alongside other issues. If you have older pipes or heavy mineral content, you may need to address multiple factors at once:
- For homes dealing with chloramine and iron, specialized Trio systems are required.
- If your water has a gritty texture, look into solutions for chloramine and sediment.

A Scientific Necessity
Understanding chlorine vs chloramine is the first step in taking control of your home’s water quality. If your city has made the switch to chloramination, a standard filter is no longer an "upgrade"; it’s an outdated tool for a modern problem. To protect your family, your plumbing, and your taste buds, a specialized chloramine system featuring catalytic carbon is a scientific necessity.
Is your city among the growing number of cities using chloramines? Find the right system for your home using our quick product quiz today.









